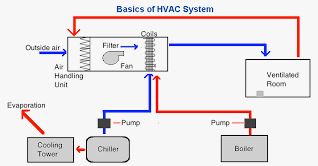
Detergents Used for Cleaning Pharmaceutical Equipment
Standard Operating Procedure (SOP) for Detergents Used for Cleaning Pharmaceutical Equipment Purpose: The purpose of this Standard Operating Procedure (SOP) is to provide guidelines for the proper and effective use of detergents in the cleaning process of pharmaceutical equipment. This SOP ensures that cleaning is carried out in a consistent and controlled manner to maintain […]
Continue Reading